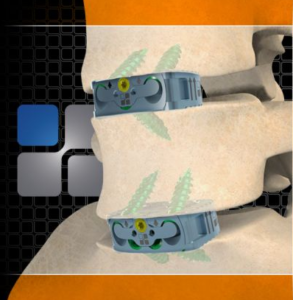
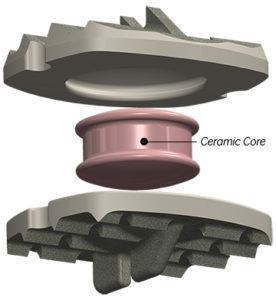
NeuroStructures is excited and pleased to announce the FDA 510k approval of the Oculus™-SA Lumbar Cage System, which features our “Neuro-Lock” surface technology to enhance fusion. The Oculus™-SA allows the surgeon to use multiple integrated screw construct options for any surgeons’ preference. Along with a zero profile design and locking plate for secure visual confirmation, […]
FDA
IZI Medical Receives CE Mark Approval for Kiva™ VCF Treatment System
OWINGS MILLS, Md., Sept. 10, 2020 /PRNewswire/ — IZI Medical Products, LLC (“IZI”), a leading manufacturer of interventional radiology devices, announces that it has received CE Mark approval in Europe for the KivaTM Vertebral Compression Fracture (VCF) Treatment System. Kiva is a unipedicular PEEK implant-based treatment solution for VCFs that has seen clinical and commercial success in the US. “We […]
Life Spine Announces FDA 510(k) Clearance for the for the PLATEAU®-A TI Anterior Lumbar Spacer System
Huntley, IL, August 5 , 2020 –Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today that it has received clearance from the U.S. Food & Drug Administration (FDA) to market the PLATEAU-A Ti Anterior Lumbar Spacer System. “With the increased usage of […]
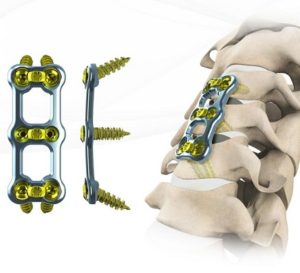
NeuroStructures is pleased to announce the FDA 510k approval of the Transept™ Cervical Plating System
NeuroStructures is excited and pleased to announce the FDA 510k approval of the Transept™ Cervical Plating System which features the ability to achieve extreme screw angulation (up to 38°) and extremely short plates (10mm). The plate has an ultra-thin profile and comes with a wide spectrum of fixed, variable, self-tapping, and self-drilling screws. Also included, […]
icotec ag Receives Approvals for Two KONG® VBR Spinal Systems Made with BlackArmor® and Ti-iT® in Europe and the US
ALTSTAETTEN, Switzerland, June 18, 2020 /PRNewswire/ — icotec ag announces that the KONG®-TL and the KONG®-C vertebral body replacement systems with the unique Titaniumcoating (Ti-iT®) receive FDA 510(k) clearance in the United States and CE approval in Europe .”This is exciting news for icotec ag and our team as it allows us to expand our portfolio in multiple countries simultaneously, to include implants […]
Meditech Spine Receives FDA Clearance for its CURE™ OPEL-L (S) system
ATLANTA, June 1, 2020 /PRNewswire/ — Meditech Spine has received FDA 510(k) clearance to market the CURE™ Opel-L (S) system, a new lumbar plate option which expands upon the previously cleared CURE™ LP Plate System and compliments its Talos®-A (HA) Interbody system. With this approval, Meditech will now offer an Interbody/Plate assembly for the anterior lumbar spine. By […]
Atlas Spine Inc., announced today the FDA 510(k) clearance and imminent launch of the V3 Guided Segmental Plating System.
JUPITER, FL, September 16, 2019 — Atlas Spine Inc., a spinal implant company based in Jupiter Florida, announced today the FDA 510(k) clearance and imminent launch of the V3 Guided Segmental Plating System. The V3 System marks another addition to the innovative portfolio of Anterior Cervical Discectomy and Fusion (ACDF) technology developed by Atlas Spine. […]
4WEB Medical Announces FDA 510(k) Clearance of its Cervical Stand-Alone Interbody Fusion Device
DALLAS, Aug. 28, 2019 /PRNewswire/ — 4WEB Medical, an orthopedic device company focused on developing innovative implants utilizing its proprietary Truss Implant Technology™, announced today that the company has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market its Cervical Spine Truss System-Stand Alone (CSTS-SA) Interbody Fusion Device. Consistent with 4WEB’s existing product portfolio, CSTS-SA has […]
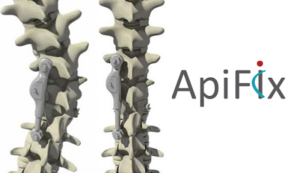
ApiFix Receives FDA Approval to Commercialize MID-C System for Motion-Preserving Deformity Treatment of Progressive Idiopathic Scoliosis in Adolescents
BOSTON–(BUSINESS WIRE)–ApiFix Ltd., a leading motion-preservation scoliosis correction company, today announced that it has received approval from the U.S. Food and Drug Administration (FDA) via a Humanitarian Device Exemption (HDE) to market the Minimally Invasive Deformity Correction (MID-C) system for the treatment of progressive adolescent idiopathic scoliosis (AIS). AIS is the most common type of […]
Nexxt Spine Adds Stand Alone Cervical to the NEXXT MATRIXX® Family
NOBLESVILLE, Ind.–(BUSINESS WIRE)–Nexxt Spine LLC, a pioneer in the design and manufacturing of innovative spinal solutions, is pleased to announce the FDA 510(k) clearance of the NEXXT MATRIXX® Stand Alone Cervical System. Born from engineering excellence, the system encompasses the cutting-edge design and surgeon friendly precision distinct to the NEXXT MATRIXX® brand of 3D printed […]
MEDICREA® Announces FDA Clearance of TULIP GENESIS to Complete its UNiDTM ASI platform technology
LYON, France & NEW YORK–(BUSINESS WIRE)–The MEDICREA® Group (Euronext Growth Paris: FR0004178572 – ALMED ; OTCQX Best Market – MRNTF), pioneering the digital transformation of spinal surgery through Artificial Intelligence, predictive modeling and patient specific implants with its UNiD™ ASI (Adaptive Spine Intelligence) proprietary software platform, services and technologies, announced today that it has received […]
Life Spine Announces FDA 510(k) Clearance of the PROLIFT® Expandable Spacer System In 8mm-10mm Widths
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today that the Food and Drug Administration has provided additional 510(k) market clearance for the PROLIFT Expandable Spacer System. “The new 8mm-10mm widths of the PROLIFT Expandable Spacer System are essential additions […]
First System for Dynamic Intra-operative Measurement of Segmental and Global Spine Alignment Receives FDA Clearance
ATLANTA, July 22, 2019 /PRNewswire/ — MiRus is pleased to announce FDA 510(k) clearance of the GALILEO™ Spine Alignment Monitoring System, a non-optical, wireless, real-time measurement system for segmental and global sagittal spine alignment. The GALILEO™ Spine Alignment Monitoring System received the 2018 Spine Technology Award at the NASS meeting for excellence and innovation in spine surgery navigation. […]
icotec AG Granted FDA Clearance To Market VADER®one Pedicle Screw System
ALTSTAETTEN, Switzerland, July 8, 2019 /PRNewswire/ — icotec AG, a Swiss company, today announced the VADER®one pedicle screw system was granted U.S. Food and Drug Administration (FDA) 510(k) clearance for both minimally-invasive and open spine surgical procedures. VADER®one, made from icotec’s unique BlackArmor® material, was designed for secure stabilization and post-operative visualization, which is important after spinal tumor procedures. In […]
Zavation Gains US FDA Clearance for the Ti3Z Interbody System (TLIF, T-PLIF, and PLIF)
FLOWOOD, Miss., June 4, 2019 /PRNewswire/ — Zavation, an employee-owned medical device company that designs, develops, manufactures and distributes medical device products, announced today that it has received 510(k) clearance from the US Food and Drug Administration (FDA) to market the Ti3Z Interbody System (TLIF, T-PLIF, and PLIF). The Mississippi company will commence launching Ti3Z Interbody system immediately to the US […]
ChoiceSpine™ Granted FDA Clearance for Two New Cervical Spinal Fusion Devices
KNOXVILLE, TENN. (PRWEB) MAY 14, 2019 –ChoiceSpine LLC, a privately-held spinal device manufacturer based in Knoxville, TN, was granted 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market TIGER SHARK C interbody device with BioBond™ technology and BOOMERANG Anterior Cervical Plate (ACP) with a two-screw configuration, for use together or separately in […]
Orthofix Announces First US Patient Implants Following FDA Approval of the M6-C Artificial Cervical Disc Designed to Treat Cervical Disc Degeneration
LEWISVILLE, Texas–(BUSINESS WIRE)–Apr. 23, 2019– Orthofix Medical Inc. (NASDAQ:OFIX), a global medical device company focused on musculoskeletal products and therapies, today announced the first commercial implants of patients with the M6-C™ artificial cervical disc. The Center for Disc Replacement at Texas Back Institute (TBI) in Dallas, Texas recently implanted four patients suffering from single level […]
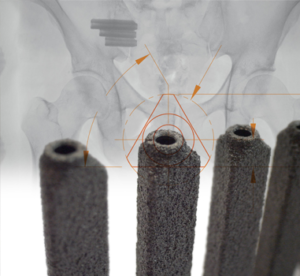
SI-BONE, Inc. Announces FDA Clearance of iFuse Bedrock™ Novel Spinopelvic Fixation Technology
SANTA CLARA, Calif., April 15, 2019 (GLOBE NEWSWIRE) — SI-BONE, Inc. (Nasdaq: SIBN), a Silicon Valley based medical device company dedicated to solving musculoskeletal disorders of the spinopelvic anatomy, today announced that it received an additional 510(k) clearance from the U.S. Food and Drug Administration (FDA) for use of its iFuse Bedrock technology in fusion […]
Collagen Matrix, Inc. Receives FDA 510(k) Clearance of a Bioactive Moldable Bone Graft Matrix
OAKLAND, N.J., April 12, 2019 /PRNewswire/ — Collagen Matrix, Inc., a leader in regenerative medicine and a global manufacturer of collagen and mineral based medical devices announced today the FDA 510(k) clearance of its innovative line of Mineral Collagen Composite Bioactive Moldable Bone Graft Matrix through its Spine business unit. OssiMend® Bioactive Moldable Bone Graft Matrix is a […]
ChoiceSpine™ Granted Expanded FDA Clearance for HAWKEYE™ Ti VBR Device
ChoiceSpine LLC, a privately-held spinal device manufacturer based in Knoxville, TN, was granted 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market HAWKEYE Ti, a 3D Printed Titanium Vertebral Body Replacement (VBR) device, for use in the cervical spine. This new indication expands the already cleared T1 to L5 indication for HAWKEYE […]
SpineEX Receives Additional FDA Clearance for Innovative Sagittae Lateral Lumbar Interbody Fusion System
FREMONT, Calif., March 25, 2019 (GLOBE NEWSWIRE) — SpineEX, Inc., a medical device company focused on the design, development and marketing of products for spine disorders, received an additional U.S. Food and Drug Administration (FDA) clearance for its innovative Sagittae Lateral Lumbar Interbody (LLIF) Fusion System. This additional 510(k) clearance will enhance SpineEX’s ability to […]
Implanet: FDA Clearance for JAZZ Cap® System
BORDEAUX, France & BOSTON: IMPLANET (Paris:ALIMP) (OTCQX:IMPZY) (Euronext Growth: ALIMP, FR0010458729, eligible for PEA-PME equity savings plans), a medical technology company specializing in vertebral and knee surgery implants, has announced that it has received 510(k) authorization from the Food and Drug Administration (FDA) for its JAZZ Cap® System, designed to meet the constraints of vertebral […]
Astura Medical Receives FDA 510(k) Clearance For OLYMPIC MIS Posterior Spinal Fixation System
CARLSBAD, CA – November 26, 2018 – Astura Medical, a high-growth, innovative spine technology company, today announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Olympic Minimally Invasive Surgery (MIS) Posterior Spinal Fixation System. The Olympic MIS system delivers a new level of intraoperative flexibility and efficiency by allowing surgeons […]
Simplify Medical Completes Enrollment in U.S. IDE Pivotal Trial of Simplify® Disc for Two-Level Cervical Disc Replacement
SUNNYVALE, Calif., Nov. 14, 2018 (GLOBE NEWSWIRE) — Simplify Medical Pty Ltd., maker of the Simplify® cervical artificial disc, today announced that it has completed the enrollment and treatment of all patients in its U.S. Investigational Device Exemption (IDE) pivotal trial evaluating the Simplify® Disc for two-level cervical disc replacement. The Simplify Disc is designed […]
VISEON, Inc. Announces FDA Clearance of the Voyant System for Minimally Invasive Spine Surgery Access, Visualization, and Illumination
IRVINE, Calif.–Viseon, Inc. today announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for their Voyant System for Minimally Invasive Spine Surgery, featuring proprietary HD imaging sensor and illumination technology. The Voyant System is composed of a sterile single-use, disposable retractor device with integrated state-of-the-art visualization technology, and a […]
K2M Receives FDA Clearance Including Surgical Guidance that Enhances MESA® Platform Using Patient-Specific Rods & Rails
LEESBURG, Va., Oct. 10, 2018 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (NASDAQ:KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today announced at the Scoliosis Research Society 53rd Annual Meeting & Course, in Bologna, Italy a U.S. Food and Drug Administration […]
SpineEX® Announces FDA Clearance of Sagittae® Lateral Lumbar Interbody Fusion Devices
FREMONT, Calif., Oct. 10, 2018 /PRNewswire/ — SpineEX, Inc., a medical device company that aims to provide innovative and minimally invasive implants, high-value disposables and instrumentation for spinal fusion surgeries, announced today it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Sagittae lateral lumbar interbody fusion (LLIF) device. The LLIF procedure […]