Éragny-sur-Oise, France, September 27, 2019 – SAFE ORTHOPAEDICS (FR0012452746 – SAFOR), a company specializing in the design and marketing of single-use implants and instruments for the minimally-invasive treatment of spine fracture pathologies, today announced its results for the first half of 2019 to June 30.
Safe Orthopaedics’ half-year financial report will be available in the Investors > Documentation > Regulated Information section of the Company’s website (www.SafeOrthopaedics.com) from September 28, 2019.
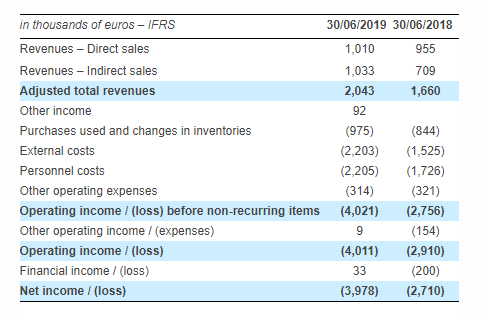
In the first half of 2019, revenues recorded strong growth of +23% compared to the first half of 2018 to €2,043 thousand, driven by indirect sales (+46%), notably thanks to the Japanese partnership. Direct sales increased by 5%. The reorganisation of the direct sales teams (FR, GER, UK) associated with the commercial launch of the new generation of the SteriSpine PS range should boost growth at the end of 2019.
This strengthening of the direct sales force in three countries in 2018, entirely borne by the first half, generated a substantial investment in the first half. Subsequently, there was a core operating loss of €4,021 thousand over the period, versus a loss of €2,756 thousand in H1 2018, and a net loss of €3,978 thousand, vs. -€2,710 thousand a year earlier.
Safe Orthopaedics had a cash position of €1.1 million at June 30, 2019, versus €474 thousand at end-June 2018. Readers are reminded that, on July 17, 2019, the Company was granted an AMF (French stock market authority) visa for its prospectus concerning a €12.15 million financing line covering its cash requirements through to 2022. The prospectus is currently being reviewed by the AMF.
“Safe Orthopaedics closed the first half of 2019 with accelerated growth in the second quarter at +32% (+16% in the first quarter) compared to 2018, thanks to the promotion of our strategic SteriSpine PS (pedicle screw combined with its ready-to-use sterile instrumentation) and SteriSpine VA (spinal augmentation balloon launched in late 2018) ranges for fracture treatment, the return to growth of distributors and our Japanese partnership,” comments Pierre Dumouchel, Chief Executive Officer and Co-founder of Safe Orthopaedics. “We also strengthened our financial visibility with the signing of a financing contract for €12.45 million, adjusted the sales forces in France, Germany and the United Kingdom to return to double-digit growth in these three countries and supported the commercial launch in Japan through our strategic partnership with Kisco. Combined with the launch of the second generation of SteriSpine PS, published on July 10, 2019, the group is working towards stronger growth in line with the trend of the first two quarters of 2019.”
Recruitment of a VP Strategy & Marketing and a Sales Director for the France & UK zones
With an Executive MBA from HEC Paris and Biomedical Engineering degree from UTC Compiègne, Sandrine Carle has over 20 years of experience in medical devices for spines. Prior to joining Safe Orthopaedics as VP Strategy & Marketing, Sandrine was Business Unit Director at Cryo Bio System. During her career, Sandrine has held a number of senior positions in marketing strategy and business development at companies such as Medtronic and Vexim.
A graduate of the ISC Paris and ESSEC business schools, Stéphane Bouchet has over 15 years of experience in Sales & Marketing within the medical device industry, six of them in the spine sector. Before joining Safe Orthopaedics as Sales Director for the France & UK zone, Stéphane held various sales and marketing management positions at Vexim, Pentax Médical, Stryker and Surgical IOC.
Next financial press release: revenues for the 3rd quarter of 2019, Thursday October 10 (after market)
About Safe Orthopaedics
Founded in 2010, Safe Orthopaedics, is a French medical technology company that offers the safest technologies to treat spinal fractures. Delivered sterile, all implants and respective disposable instrumentation are available to the surgeon at any time, anywhere. These technologies enable minimally invasive approaches, redcucing risks of cross contamination and infection in the interest of the patient. Protected by 17 patent families, SteriSpineTM kits are CE marked and FDA cleared. The company is based in Eragny-sur-Oise (95) and has 40 employees.
For more information: www.SafeOrthopaedics.com
