WARSAW, Ind., Oct. 16, 2017 (GLOBE NEWSWIRE) — OrthoPediatrics Corp. (NASDAQ:KIDS), an orthopedic company focused exclusively on providing a comprehensive product offering to the pediatric orthopedic market, announced today the closing of its initial public offering of 4,600,000 shares of its common stock, including the full exercise by the underwriters of their option to purchase […]
NEWS
NuVasive Receives Expanded FDA 510(k) Clearance For TLX Interbody System Used In TLIF Procedures
SAN DIEGO, Oct. 16, 2017 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), a leading medical device company focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced an expanded U.S. Food and Drug Administration (FDA) 510(k) clearance of the Company’s TLX interbody system, used in the leading spinal fusion surgery. New clearance introduces an […]
Spine Surgeons in Texas and Indiana Advance Patient Care with Structurally Encoded Devices
Thursday, October 12, 2017–Zeshan Hyder, DO, of Bone & Joint Specialists in Merrillville, Ind.; Frank K. Kuwamura, III, MD, of Innovative Spine in San Antonio; and Larry M. Kjeldgaard, DO, of Cervical Spine Specialist in Fort Worth are the first surgeons to implant medical devices with Structural Encoding® technology. The Structural Encoding technology advances patient […]
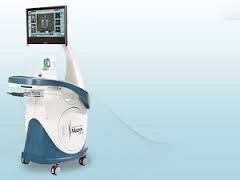
Mazor Robotics Appoints Medical Device Industry Veteran Ron Tavlin as Vice President- Business Development
CAESAREA, Israel–(BUSINESS WIRE)–Mazor Robotics Ltd. (TASE: MZOR; NASDAQGM: MZOR), a pioneer and a leader in the field of surgical robotic guidance systems, announced the appointment of Ron A. Tavlin as Vice President, Business Development. In this newly created role, Mr. Tavlin will report directly to Ori Hadomi, Mazor’s Chief Executive Officer. “Last year, we began […]
NuVasive Receives Expanded FDA 510(k) Clearance For Innovative Magnetic Limb Lengthening Technology
SAN DIEGO, Oct. 12, 2017 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), a leading medical device company focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced that it has received expanded 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the Company’s PRECICE® system from NuVasive Specialized Orthopedics™ (NSO) with expanded […]
Alphatec Denounces “Baseless” NuVasive Complaint
CARLSBAD, Calif., Oct. 11, 2017 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (Nasdaq:ATEC), a provider of innovative spine surgery solutions with a mission to improve patient lives through the relentless pursuit of superior outcomes, released a statement in response to a recent complaint filed by NuVasive, Inc. (Nasdaq:NUVA) against Patrick S. Miles, Alphatec’s recently appointed Executive […]
Nuvasive Files Lawsuit Against Patrick S. Miles To Protect Corporate Assets And Stakeholders’ Interests
SAN DIEGO, Oct. 10, 2017 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), a leading medical device company focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced that it has filed a lawsuit in the Delaware Chancery Court against Patrick S. Miles, former vice chairman of the Company and a member of NuVasive’s Board of Directors. The lawsuit asserts […]
Globus Medical Announces First Spine Surgeries Using ExcelsiusGPS™
AUDUBON, Pa., Oct. 10, 2017 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced the first spine surgeries using ExcelsiusGPS™, a revolutionary robotic guidance and navigation system. Surgeries using ExcelsiusGPS™ were performed last week at The Johns Hopkins Hospital in Baltimore, Maryland and St. Mark’s Hospital in Salt Lake City, […]
Xtant Medical to shut down Dayton, Ohio facility
Xtant Medical (NYSE:XTNT) plans to close its Dayton, Ohio facility and transition its operations to the company’s headquarters in Belgrade, Montana. The Dayton facility employs 55 people in an array of quality assurance, regulatory, inventory management, finance, engineering and distribution jobs, the company said, adding that many of these functions will transition to the Montana facility. […]
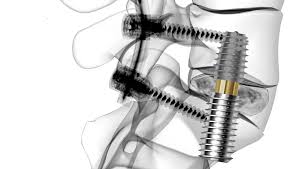
Expandable interbody cages: Are they here to stay? Learn about the 27 most relevant devices
Will Expandable interbody cages succeed in the spinal market? According to many surgeons and many of the Spinal companies, these new devices may help surgeons improve efficiency in the operating room and increase patient safety during TLIF, PLIF and even Lateral cages procedures. These advantages are the following: Expandable interbody cages allow to the surgeon to […]
Safe Orthopaedics: Very Strong Revenue Growth in Q3 2017: +63%
ERAGNY-SUR-OISE, France–Regulatory News: SAFE ORTHOPAEDICS (Paris:SAFOR) (FR0012452746 – SAFOR), a company offering an innovative range of sterile implants combined with their single-use instruments for spinal surgery, is today announcing its revenues for the third quarter of 2017 and its cash position at September 30, 2017. Thousands of euros Q3 2017 Q3 2016 Change France 336 […]
Implanet: 20% Increase in Revenue In Q3 2017.30% growth in Jazz® sales
BORDEAUX, France & BOSTON–Regulatory News: IMPLANET (Paris:IMPL) (OTCQX:IMPZY) (Euronext Growth: ALIMP, FR0010458729, PEA-PME eligible), a medical technology company specializing in vertebral and knee-surgery implants, today announces its sales for the third quarter and first nine months to September 30, 2017. Ludovic Lastennet, CEO of Implanet, says: “The Group’s activity was solid last quarter. The increase […]
Stryker’s 3D-Printed Tritanium® C Anterior Cervical Cage To Be Featured at Congress of Neurological Surgeons Meeting
ALLENDALE, N.J.–(BUSINESS WIRE)–Stryker’s Spine division will demonstrate its new 3D-printed Tritanium C Anterior Cervical Cage, an interbody fusion device designed for use in the cervical spine, at the Congress of Neurological Surgeons (CNS) Annual Meeting, Oct. 7-11, 2017, in Boston (booth No. 311). The Tritanium C Anterior Cervical Cage is the latest spinal fusion implant constructed […]
AxioMed Opens $10 Million Second Round to Fund Multi-Level Cervical IDE and Expand Commercialization
MALDEN, Mass.–(BUSINESS WIRE)–AxioMed closed an oversubscribed first round of funding from surgeons and high net worth individuals. Its second round for $10 million currently has pledges prior to the planned launch in October. “The momentum from the first round and strong interest for the second round secures AxioMed operations and allows it to aggressively pursue […]
Xtant Medical Announces Consolidation of Fixation Operations to Montana and Closure of Dayton Facility
BELGRADE, Mont., Oct. 06, 2017 (GLOBE NEWSWIRE) — Xtant Medical Holdings, Inc. (NYSE American:XTNT), a leader in the development of regenerative medicine products and medical devices, today announced the closure of its Dayton, Ohio facility and the transitioning of its fixation operations to the Company’s headquarters in Belgrade, Montana. This decision will allow the Company […]
Spineguard Reports €6.0m Revenue and 13% Growth for the 9 Months of 2017, US Third-Quarter Growth of 23% cc
PARIS & SAN FRANCISCO–(BUSINESS WIRE)–Regulatory News: SpineGuard (Paris:ALSGD) (FR0011464452 – ALSGD), an innovative company that designs, develops, and markets disposable medical devices intended to make spine surgery safer, announced today that its 9 months revenue grew to €6.0 million, a 13% increase compared with the same period in 2016. Stéphane Bette, CEO and co-founder of […]
K2M Group Holdings, Inc. President & Chief Executive Officer Eric Major Elected Chairman of the Board of Directors
LEESBURG, Va., Oct. 04, 2017 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (NASDAQ:KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today announced that President and Chief Executive Officer Eric Major has been elected Chairman of the Company’s Board of Directors, effective immediately. Major […]
ChoiceSpine™ Granted FDA Clearance for 3D Printed Vertebral Body Replacement Device
KNOXVILLE, TN (PRWEB) OCTOBER 04, 2017-ChoiceSpine LP, a privately-held spinal device manufacturer based in Knoxville, TN, announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market HAWKEYE Ti, a 3D Printed Titanium Vertebral Body Replacement (VBR) device. “Additive manufacturing techniques create intricate three-dimensional implants, layer by layer, allowing a […]
4WEB Medical Announces Launch of Next Generation Anterior Spine Truss System
DALLAS, Oct. 4, 2017 /PRNewswire/ — 4WEB Medical, the industry leader in 3D printed implant technology, has announced the launch of its next generation interbody fusion product line for anterior lumbar spine procedures. The new release of the Anterior Spine Truss System recently received FDA clearance for several impactful line extensions along with some key new indications for […]
Nexxt Spine Announces FDA Clearance for the NEXXT MATRIXX™ System
NOBLESVILLE, Ind.–(BUSINESS WIRE)–Nexxt Spine, LLC, a medical device company focused on designing, manufacturing, and distributing innovative spinal solutions, today announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market the NEXXT MATRIXX™ System. The NEXXT MATRIXX™ System of 3D printed porous titanium leverages Nexxt generation technology to create […]
Zimmer Biomet Announces U.S. Launch of Avenue® T TLIF Cage with Integrated VerteBRIDGE® Plating
WARSAW, Ind., Oct. 2, 2017 /PRNewswire/ — Zimmer Biomet Holdings, Inc. (NYSE and SIX: ZBH), a global leader in musculoskeletal healthcare, today announced it is officially launching in the United States the Avenue® T TLIF Cage. Avenue T advances posterior lumbar cage technology by incorporating VerteBRIDGE® plating, which facilitates simplified cage insertion and zero-profile, intradiscal fixation through a direct, minimally invasive surgical […]
Medicrea Presents Patient-Specific UNiD™ Rod Clinical Results Showing Very Significant Reduction in Rod Breakage
LYON, France & NEW YORK–(BUSINESS WIRE)–The Medicrea Group (Euronext Growth Paris: FR0004178572 – ALMED), pioneering the convergence of healthcare IT and next-generation, outcome-centered device design and manufacturing with UNiD™ ASI technology, announced today the results of a new White Paper titled Patient-Specific Rods show a reduction in rod breakage incidence. The paper shows that, relative to […]
Alphatec Accelerates Business Transformation with Additional Spine-Experienced Leadership Appointments
CARLSBAD, Calif., Oct. 02, 2017 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (Nasdaq:ATEC), a provider of innovative spine surgery solutions with a mission to improve patient lives through the relentless pursuit of superior outcomes, announced that Patrick Miles has been appointed as Executive Chairman and Quentin Blackford as a member of the Board, effective today. Miles […]
Europe Spinal Implants Market is Projected to Reach US$ 3 Billion by 2024
According to the new report published by Market Research Engine, the Europe Spinal Implants Market is expected to exceed more than US$ 3.8 Billion by 2024 at a CAGR of 5.7% in the given forecast period. Related to their new publication titled as “Europe Spinal Implants Market By Product Analysis (Cervical Fusion Devices, Spine Biologics, Non-fusion […]
K2M Receives FDA Clearance for Next-Generation YUKON™ OCT Spinal System
LEESBURG, Va., Sept. 29, 2017 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (NASDAQ:KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today announced that it has received 510(k) clearance from the U.S. Food & Drug Administration (FDA) for its YUKON™ OCT Spinal System. YUKON OCT offers […]
Journal of Comparative Effectiveness Research Publishes Pivotal Level 1 Evidence Comparing activL® Artificial Disc to ProDisc-L, Fusion and Conservative Care
CENTER VALLEY, Pa., Sept. 28, 2017 /PRNewswire/ — Aesculap Implant Systems, LLC and Cornerstone Research Group, Inc. today announced the publication of a Network Meta-Analysis (NMA) which was conducted to measure the varying impacts of different treatment modalities for single-level lumbar Degenerative Disc Disease (DDD). The NMA results show activL Artificial Disc is more likely to address patient […]
TranS1® and CU Innovations Partner to Commercialize Ideas Originating at Anschutz Medical Campus
Denver, Sept. 28, 2017 (GLOBE NEWSWIRE) — TranS1®, a med-tech company creating advanced surgical options for better pathways to patient care, announced today a partnership with CU Innovations and Orthopedic Surgery at the University of Colorado Anschutz Medical Campus. CU Innovations works directly with CU researchers to bring their innovative ideas and concepts to the marketplace, to […]