PLEASANTON, Calif., Sept. 20, 2018 /PRNewswire/ — Providence Medical Technology, Inc., an innovator in tissue-sparing surgical equipment and implants for cervical spine fusion surgery, today announced the closing of $25 million in new equity financing. Revelation Partners led the round with participation from the private equity investment team at BMO Global Asset Management (EMEA), MVM […]
NEWS
Stryker’s Spine Division To Showcase Tritanium® In-Growth Technology in Augmented Reality Experience at NASS 2018
ALLENDALE, N.J.–(BUSINESS WIRE)–Stryker’s Spine division will feature its family of 3D-printed Tritanium interbody fusion cages and highlight its Tritanium In-Growth Technology1 in an augmented reality experience at the North American Spine Society Annual Meeting, Sept. 26–29, 2018, in Los Angeles (booth No. 1401). The augmented reality “tour” combines objects in the real world with computer-augmented […]
Medicrea Announces Eurospine 2018 Presence and Highlights a Recently Published Study Validating Its Predictive Modelling Approach for AIS Patients
LYON, France & NEW YORK–(BUSINESS WIRE)–The Medicrea Group (Euronext Growth Paris: FR0004178572 – ALMED, PEA-PME eligible, and OTCQX: MNRTY and MNRTF), pioneering the transformation of spinal surgeries through Artificial Intelligence, predictive modeling and patient specific implants with its UNiD™ ASI (Adaptive Spine Intelligence) technology, announced today that the Company is attending the Eurospine 2018 meeting […]
joimax® Introduces Intracs® em Electromagnetic Navigation System
KARLSRUHE, Germany & IRVINE, Calif.–(BUSINESS WIRE)–joimax®, the Germany-based market leader of technologies and training methods for full-endoscopic spinal surgery, introduces another minimally invasive device, Intracs® em Electromagnetic Navigation System, at two global spine events: EUROSPINE Barcelona, the Spine Society of Europe’s annual conference and expo (September 19-21), and NASS 2018, the North American Spine Society […]
Medicrea Reports First Half 2018 Results
LYON, France & NEW YORK–(BUSINESS WIRE)–The Medicrea Group (Euronext Growth Paris: FR0004178572 – ALMED, PEA-PME eligible, and OTCQX: MNRTY and MNRTF), pioneering the transformation of spinal surgeries through Artificial Intelligence, predictive modeling and patient specific implants with its UNiD™ ASI (Adaptive Spine Intelligence) technology, has today published its unaudited results for the first half of […]
Safe Orthopaedics announces the commercial launch of SteriSpineTMVA, a balloon for the Vertebral Augmentation
Eragny-sur-Oise, France, September 18, 2018, 5:45pm CEST – SAFE ORTHOPAEDICS (FR0012452746 – SAFOR), a company offering innovative ranges of sterile implants combined with their single-use instruments for back surgery, today announces its worldwide launch of its Kyphoplasty System: the SteriSpineTM VA. The new system adds to an already existing portfolio of sterile & single use devices […]
SpineGuard Reports Six-Month 2018 Financial Results
PARIS & SAN FRANCISCO–(BUSINESS WIRE)–Regulatory News: SpineGuard (FR0011464452 – ALSGD) (Paris:ALSGD), an innovative company that designs, develops, and markets disposable medical devices intended to make spine surgery safer by bringing real-time digital technology into the operating room, announced today financial results for the half year ending June 30, 2018, as approved by the Board of […]
Life Spine Announces Record Sales Growth of SIMPACT® Sacroiliac Fixation System
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today continued accelerated market adoption and high sales growth for the SIMPACT Sacroiliac Joint Fixation system, since commercialization at the start of the 2018 calendar year. “Clinical economics and patient outcomes are […]
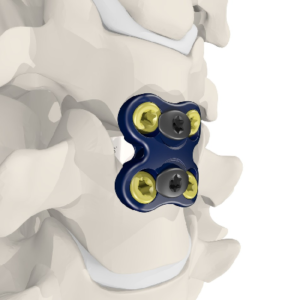
Meditech Spine Receives FDA Clearance for its Cure™ Opel-C Plating System
ATLANTA, Sept. 18, 2018 /PRNewswire/ — Meditech Spine has received FDA 510(k) clearance to market the Cure™ Opel-C Plate System, an add-on technology to its already cleared CURE™ ACP System and Talos®-C (HA) Interbody Systems. With this approval, Meditech will now offer two additional CURE™ ACP plating options with its Talos® line of cervical interbodies made with the […]
Zavation Medical Products, LLC, a LongueVue Capital Portfolio Company, Completes Investment in Pan Medical U.S. Corp.
JACKSON, Miss., Sept. 18, 2018 /PRNewswire/ — Zavation Medical Products (“Zavation”), a LongueVue Capital (“LVC”) portfolio company, is pleased to announce it has partnered with the management team of Pan Medical U.S. Corp (“PanMed” or the “Company”) to acquire the Company. This partnership broadens Zavation’s service offering by adding a full suite of minimally invasive products, including the […]
TranS1® Announces NICE Guidance for Approval of AxiaLIF® in the UK’s National Health System (NHS)
WILMINGTON, N.C., Sept. 18, 2018 /PRNewswire/ — TranS1®, an innovator in spine technology development that pioneered the anterior retroperitoneal presacral approach utilizing the AxiaLIF® System, announced today that the UK’s National Institute for Health and Care Excellence (NICE) published its Interventional Procedure Guidance recommendation for transaxial interbody lumbosacral fusion for severe chronic low back pain.1 The guidance recommended that […]
SpineVision® Receives CE Mark for Next-Gen P.L.U.S.® Deformity-Correction Spinal System
ANTONY, France–(BUSINESS WIRE)–SpineVision announced today that it has received CE mark for its next-generation Pivot Link Universal System (P.L.U.S.) spine deformity-correction system. SpineVision will introduce the next-generation P.L.U.S. to spine surgeons at “EuroSpine 2018” (Booth #24C) in Barcelona, Spain (Sept. 19-21), and “NASS 2018” (Booth #1311) in Los Angeles (Sept. 26-29). Deformity of the spine includes any abnormality of the formation, alignment, or shape of the vertebral […]
BioMedGPS Launches SmartTRAK Robotic and Computer Assisted Surgery – Spine Module
IRVINE, CALIF. (PRWEB) SEPTEMBER 18, 2018–BioMedGPS announces expanded coverage of SmartTRAK Business Intelligence in the realm of Robotic and Computer Assisted Surgery with the launch of its first module, US Computer Assisted Surgery (CAS) – Spine at the upcoming NASS conference later this month. A second SmartTRAK module, US Computer Assisted Surgery (CAS) – Ortho, will […]
ImplantBase Growth Accelerates with Digital Transformation Imperative
AUSTIN, TEXAS (PRWEB) SEPTEMBER 18, 2018–2018 has all the makings of a banner year for ImplantBase, the creator of the industry-leading, cloud-based software platform that drives digital transformation for orthopedic implant manufacturers. In addition to record growth in new implementations of its platform, the company has unveiled a refresh of its brand and digital presence. Amid […]
MiRus™ Wins Two 2018 Spine Technology Awards
ATLANTA, Sept. 17, 2018 /PRNewswire/ — MiRus has won the Annual Spine Technology Awards for both of its initial products in the implant and navigation categories. The Spine Technology Awards are intended to bring increased recognition to exemplary and innovative spine surgery products and the engineering teams and inventors who create them. The awards will be presented to […]
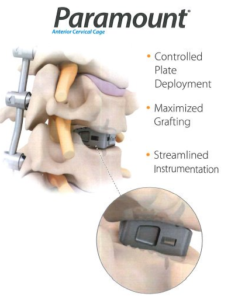
Spine Wave Announces the Commercial Launch of the Paramount® Anterior Cervical Cage
SHELTON, Conn., Sept. 17, 2018 (GLOBE NEWSWIRE) — Spine Wave is pleased to announce the commercial launch of the Paramount® Anterior Cervical Cage. The Paramount® Anterior Cervical Cage is a titanium anterior cervical implant with integrated fixation blades. The patented system offers controlled, less invasive blade deployment and maximized bone grafting. The Paramount® Anterior Cervical […]
Simplify® Medical Technology Selected for Innovative Technology Paper at NASS 2018 Annual Meeting
SUNNYVALE, Calif., Sept. 17, 2018 (GLOBE NEWSWIRE) — Simplify Medical Pty Ltd, maker of the Simplify® cervical artificial disc, announced today that an abstract for preliminary data from the Company’s 1-level Investigational Device Exemption (IDE) clinical trial has been selected as an Innovative Technology Presentation during the 2018 North American Spine Society (NASS) Annual Meeting, […]
SpineVision® Receives FDA Clearance and CE Mark for Next-Gen Titanium 3D-Printed HEXANIUM TLIF Cage
ANTONY, France–(BUSINESS WIRE)–SpineVision announced today that it has received FDA clearance and CE mark for its next-generation titanium 3D laser-printed HEXANIUM TLIF (Transforaminal Lumbar Interbody Fusion) cage for back surgery. SpineVision will introduce its new product to spine surgeons at “EuroSpine 2018” (Booth #24C) in Barcelona, Spain (Sept. 19-21), and “NASS 2018” (Booth #1311) in […]
Globus Medical Acquires Surgimap®
AUDUBON, Pa., Sept. 13, 2018 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced the acquisition of Nemaris Inc., a privately held company that markets and develops Surgimap®, a leading surgical planning software platform. Surgimap® allows healthcare professionals to simulate potential surgical outcomes and share medical imaging globally to improve […]
SpineGuard Secures 9.0 M€ Financing
PARIS & SAN FRANCISCO–(BUSINESS WIRE)–Regulatory News:SpineGuard (Paris:ALSGD) (FR0011464452 – ALSGD), an innovative company that develops and markets instruments designed to secure the placement of surgical implants by bringing real-time digital technology into the operating room, announced today that it secured 9.0 million euro of financing. The two financings consist of: The issuance of a 6.0 million […]
Life Spine Announces Initial Cases of NAUTILUS® Thoracolumbar System at Shriners Hospital in Mexico City
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today first clinical cases of the Life Spine’s NAUTILUS Thoracolumbar Spinal Fixation System with Dr. Jose Antonio Canales and Dr. Antonio Hurtado Padilla, surgeons specializing in deformity correction at Shriners Hospital, Mexico […]
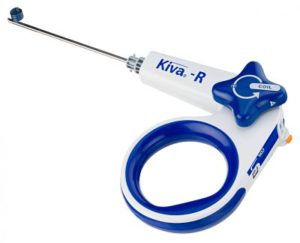
IZI Medical Products Acquires Benvenue Medical’s Vertebral Augmentation Systems
OWINGS MILLS, Md.–(BUSINESS WIRE)–IZI Medical Products, LLC, a leading interventional medical device company, announced it has completed the acquisition of Benvenue Medical, Inc.’s vertebral augmentation systems. The acquisition includes the Kiva®, Kiva® Pilot, and Blazer® devices in addition to other needles, injectors and cements. This acquisition complements IZI Medical Products’ vertebroplasty and bone biopsy lines […]
Benvenue Medical Completes Divestiture of its Vertebral Augmentation Systems Portfolio
SANTA CLARA, Calif.–(BUSINESS WIRE)–Benvenue Medical, Inc., a developer of minimally invasive expandable implant solutions for lumbar fusion, today announced it has completed the divestiture of its vertebral augmentation systems portfolio for an undisclosed amount to IZI Medical Products, LLC. The completion of the sale of this business allows Benvenue Medical to devote its full resources […]
Captiva Spine Receives Clearance to Market TowerLOX-EXT MIS Extended Tab Pedicle Screw
JUPITER, FLA. (PRWEB) SEPTEMBER 12, 2018–Captiva Spine, Inc., which designs, manufactures and distributes elegant and intuitive spine fusion solutions announced today it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market TowerLOX-EXT® MIS Extended Tab Pedicle Screw complementing its TowerLOX® MIS Pedicle Screw System. TowerLOX-EXT MIS Extended Tab Pedicle Screws provide […]
Johnson & Johnson Medical GmbH Acquires Emerging Implant Technologies GmbH to Enhance Global Offering of Interbody Spine Implants
NORDERSTEDT, Germany, Sept. 12, 2018 /PRNewswire/ — Johnson & Johnson Medical Devices Companies*, through its subsidiary Johnson & Johnson Medical GmbH, announced today the acquisition of Emerging Implant Technologies GmbH (EIT), a privately held manufacturer of 3D-printed titanium interbody implants for spinal fusion surgery, based in Wurmlingen, Germany. The products in this portfolio leverage EIT’s […]
CoreLink Surgical, LLC, Today Announced 510(k) Clearance to Market the M3™ Stand-Alone Anterior Lumbar (ALIF) System
ST. LOUIS–(BUSINESS WIRE)–CoreLink Surgical, LLC, today announced 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market the M3™ Stand-Alone Anterior Lumbar (ALIF) System. Jay Bartling, CEO, said, “M3 is our first stand-alone interbody fusion device to feature 3D printing – I continue to be impressed by the ability of our design teams […]
Nanovis Licenses Key Nanosurface Technology Patent from the University of Nevada, Reno
CARMEL, IND. (PRWEB) SEPTEMBER 11, 2018//Nanovis, a leader in nanomedicine for the spine, today announced a licensing agreement with the University of Nevada, Reno for a key nanosurface technology patent covering the use of ceramics on implants with nanopores. This foundational patent allows development of ceramics for medical implants with nanosurfacing that enhances cell binding […]